Article by: Mr. Larry Jay B. Langaman, BRAP Asst. Auditor
Edited by: Mr. Ariel Angelo B. Aquino, BRAP Asst. Secretary
Six years after the association successfully established footing, the BioRisk Association of the Philippines 2015, Inc. (BRAP2015) recalibrated its focus as it underwent a 2-month strategic planning process amidst the unprecedented yet simplifying distortion of the COVID-19 pandemic. Incumbent BRAP officers, despite their distance and busy schedules, actively engaged in a carefully phased strategic thinking process on how the association repositions itself as it takes wider reach while creating value for its members and the causes it represents.
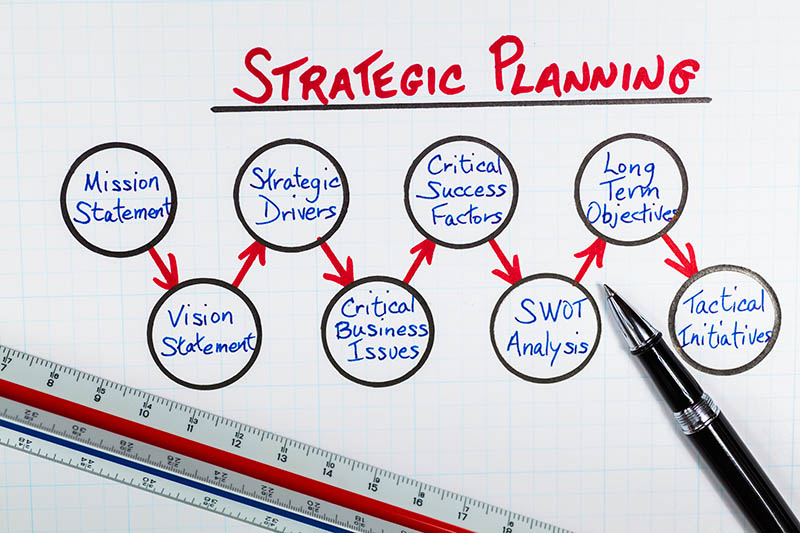
BRAP2015 strategic planning went through three phases that commenced with the mission–vision review, strengths, weaknesses, opportunities, and threats analysis, and identification of key priority areas (KPAs) based on the result of organizational internal and external scanning (figure 1). Phase 2 processed the inputs from phase 1 through in-depth discussion, challenging positions and projection, and ranking of processed KPA. Phase 3 grouped previously ranked KPA and packaged into a cohesive, clear, and intact message, which was presented and accepted by the board in April.

The reliable planning inputs came from the nine incumbent BRAP officers (figure 2) who are also current leaders and prime movers of local and international organizations, that is, key decision makers of various sectors of healthcare and academic industries. The process generated a wealth of insights that served as a springboard for BRAP’s repositioning tool.

After 2 months of multiple online sessions and asynchronous individual quality strategic thinking activities, three BRAP KPAs surfaced: organizational development, advocacy and reputation (figure 3). The planning team unanimously agreed that organizational development is a prerequisite to sustainable advocacy and an excellent organizational reputation. Organizational development will focus on the improvement of membership services, financial stability, and the development of new and upcoming leaders who will champion biosafety and biosecurity. Advocacy is the heartbeat of BRAP2015 zeroing in on the conduct of educational programs and promotion of biosafety and biosecurity culture. Reputation builds on the former KPAs as it takes the ambitious responsibility of becoming a certifying body for biosafety and biosecurity, which is a product of long-term network and collaboration with local and international organizations. In an effort to validate 360-degree coverage of BRAP2015’s strategic planning output, BRAP’s newly crafted KPAs and goals were assessed against and found to be compliant with four areas of Kaplan’s Balanced Scorecard model: customer, internal business process, financial, and learning and growth.

BRAP2015 is set to operationalize its strategic plan through its committees, each tasked to set key performance indicators, leading, and lagging metrics. BRAP’s founding and re-elected president, Dr. Miguel Martin N. Moreno II, signals the transition of the association from a necessary central authoritative style in order for the association to take-off in 2015, to a more permissive and pacesetting approach with all rights transferred to the Board. He envisions the next years of BRAP2015 with empowered leaders and officers who are accountable and result driven in strengthening the association’s resources, reach, and reputation in the coming years.
About the author: Larry Jay B. Langaman is a medical technologist certified by PRC and ASCP. He held management positions in various ancillary units of tertiary healthcare institution prior to his early retirement. He currently manages a water testing laboratory in Davao City and a faculty member of higher education institutions in Regions XI and XII. He is actively engaged with various government and non-government organizations such as Philippine Association of Medical Technologists, Inc., Department of Health’s Regional Lab Network and as BRAP’s incumbent assistant auditor. Prior to joining BRAP, he has been actively supporting biosafety and biosecurity advocacies in Mindanao. Knowing Sir Larry’s mastery of the strategic planning process, Dr. Moreno tasked Mr. Langaman to spearhead the 2021–2025 strategic planning.
Trivia QUIZ link http://bit.ly/BRAPStratPlanQuiz
Good Luck













